CareStartTM COVID-19 Antigen
Rapid Diagnostic Test for the Detection of SARS-CoV-2 Antigen
CareStart™ COVID-19 Antigen Rapid Point-of-Care Test*
The CareStart™ COVID-19 Antigen Test is a lateral flow immunochromatographic assay for the detection of extracted nucleocapsid protein antigens specific to SARS-CoV-2 in swab specimens directly collected from individuals who are suspected of COVID-19 by their healthcare providers.
As an intended point-of-care (POC)* designated test with results to be read at 10 minutes, CareStart™ COVID-19 Antigen Test allows diagnostic indication of COVID-19 by qualified healthcare professionals on a large scale.

* This test is authorized for point-of-care use by users with a CLIA certificate of wavier.
Access Bio CareStart COVID-19 Rapid Antigen Test
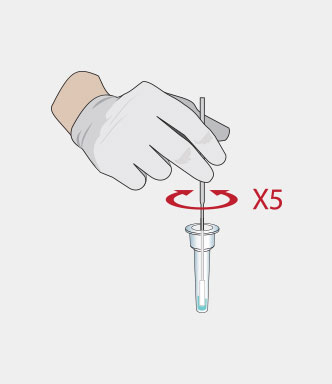
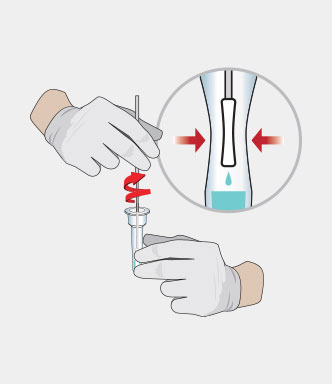
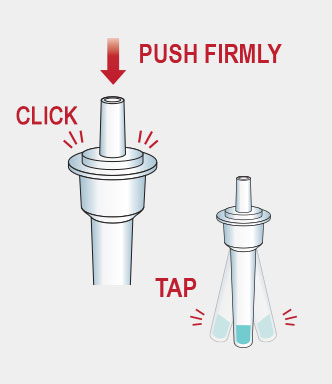
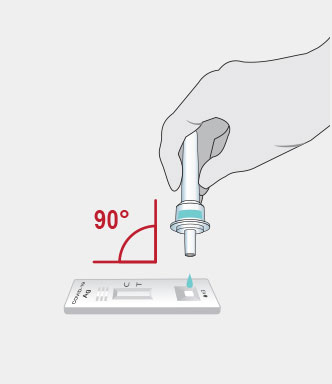
Procedure
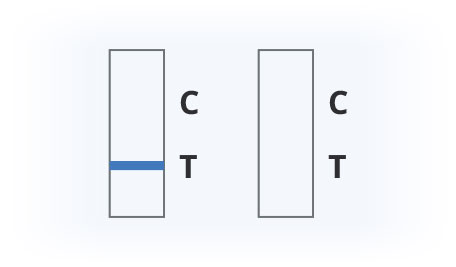
Results Interpretation
Covid-19 Positive
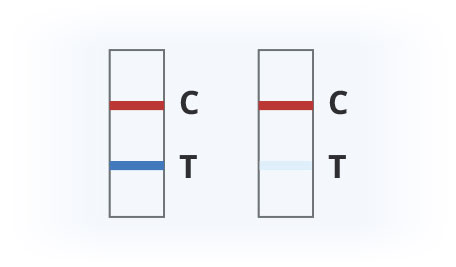
Covid-19 Negative
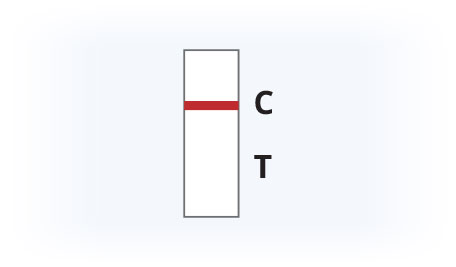
Invalid